Inorganic Chemistry
Uncategorized
Metals and Non Metals Class 10 Notes PDF download, Metals and Non Metals Class 10 Notes Prashant Sir 1, Metals and Non Metals Class 10 Notes self study, Metals and Non Metals Notes PDF, Metals And Non-Metals Complete Chapter Notes With Examples
sadrealam.umg@gmail.com
1 Comments
Metals And Non-Metals Complete Chapter Notes With Examples
Metal
Those substance which can lose electrons are called metals. They are generally found on the side of the periodic table.
Non- Metal
Those Substance which gain electron to form negative ions are called non-metals. They are found on the side of the periodic table.
Metalloid
Elements which show properties of both metals and non-metals are called metalloids.
Example- Silicon (Si)
Difference Between Metals And non-metals
| Metals | Non-Metals |
|---|---|
| Metals are generally Solid in Nature. Like Na, Mg, Ca etc. | Non-Metals generally soft. |
| Metals generally solid in nature. | Non-Metals May be Solid, Liquid or Gas. |
| Metals are shiny in appearance. | Non-Metals are dull in appearance. |
| Metals are good conductors of heat and Electricity. | Non-Metals are poor conductors of heat and Electricity. |
| Metal have high melting point. | Non-Metal have low melting point. |
| Metals have 1,2 or 3 Electron in their outermost shell. | Non-Metals have 4,5,6 or 7 Electron in their outermost shell. |
| Metals easily lose electrons. | Non- Metals gain electrons easily. |
| Metals react with water to form metal hydroxide and hydrogen gas. | Non- Metals generally do not react with water. |
Reactivity Series
Arrangement of metals in decreasing order of their reactivity is called the reactivity series.
Reactivity Series (High to Low)- K>Na>Ca>Mg>Al>Zn>Pb>H>Cu>Hg>Ag>Au
Potassium is the most reactive metal, while Gold is the least reactive metal.
| Metal Name | Symbol | Reactivity |
|---|---|---|
| Potassium | K | Very High |
| Sodium | Na | Very High |
| Calcium | Ca | High |
| Magnesium | Mg | Medium |
| Aluminium | Al | Medium |
| Zinc | Zn | Medium |
| Lead | Pb | Medium |
| Hydrogen | H | Reference |
| Copper | Cu | Low |
| Mercury | Hg | Low |
| Silver | Ag | Very Low |
| Gold | Au | Least |
Chemical Properties Of Metals
1 Reaction of metals with air
- Metals react with oxygen present in air to form metal oxides.
Metal + O (Oxygen)——-> Metal Oxide
- Sodium (Na) and Potassium (K) react very fast with air and catch fire easily. Therefore, to prevent them from reacting , they are stored in kerosene oil.
4Na + O2 —–> 2NaO
- Some Metals like ( Mg, Al, Zn & Pb) react slowly with air. Due to this reaction, a thin layer of oxide is formed on their surface. This oxide layer is useful for these metals because it is hard and prevents further contact of the metal with air.
4Al + 3O2 ——> 2Al2O3
2Mg + O2 ——> 2MgO
- Iron (Fe) does not react with air even on heating, but when iron powder is heated strongly, it reacts rapidly with air.
- Silver(Ag) and Gold(Au) do not react with oxygen.
Amphoteric Oxides Definition
Those metal oxides which react with both acids and bases to form salt and water are called amphoteric oxides.
Al2O3 + 6HCl —> 2AlCl3 + H2O
Al2O3 + 2NaOH —-> 2NaAlO2 + H2O
2 Reactions Of Metals With Water
When metals react with water, they form metal hydroxide ( Metal oxide) and hydrogen gas. Highly reactive metals such as sodium, potassium and calcium react with cold water.
Sodium (Na) Reacts with water to form Sodium Hydroxide (NaOH) and Hydrogen (H) gas.
2Na + H2O —-> 2NaOH + H2
Calcium (Ca) Reacts with Water to form Calcium Hydroxide (CaOH) And Hydrogen gas.
Ca + H2O —–> Ca(OH)2 + H2
Magnesium(Mg) React With Hot Water.
Mg + 2H2O ——> Mg(OH)2 + H2
Calcium and Magnesium React with Water, they start floating on the surface of water. This happens because during the reaction, bubbles of hydrogen gas are produced, which stick to the surface of the metal and make it float.
Al, Fe And Zn are less reactive metals. They react only with steam.
2Al + 3H2O (Steam) ——-> Al2O3 + 3H2
2Fe + 4H2O (Steam) ——> Fe3O4 + 4H2
Cu, Al, Ag, Au And Si do not react with water.
Reaction of Metals With Dilute Acids Complete Explanation
Highly reactive metals react with dilute acids. During this reaction, Salt and Hydrogen gas are formed.
Metal + Dilute —-> Salt + Hydrogen gas
2Na + 2HCl —-> 2NaCl + H2 (gas)
Mg + 2HCl —-> MgCl2 + H2 (gas)
Zn + 2HCl —-> ZnCl2 + H2 (gas)
2Al + 6HCl —-> 2AlCl3 + 3H2 (gas)
Note- Metals that are below hydrogen in the reactivity series do not react with dilute Acids.
Reaction of Metals with Other Metals ( Displacement Reaction)
A more reactive metal displaces a less reactive metal form its compound solution. This type of reaction is called a Displacement Reaction.
Metal (A) + Salt Solution (B) ——-> Salt Solution Of Metal (A) + Metal (B)
Reaction of Metals with Non-Metals
When a metal reacts with a non-metal, the atoms of the metal lose their electrons and form positive ions (cations). On the other hand, the atoms of the non-metal gain these electrons and form negative ions (anions). The positively charged ion of the metal and the negatively charged ion of the non-metal combine together to form an ionic compound. The bond formed between two atoms due to the transfer of electrons is called an ionic bond.
Example – Sodium chloride (NaCl), Potassium chloride (KCl)
Properties of Ionic Compounds
Physical Nature
Ionic compounds are generally solids.
This is because the force of attraction between their ions is very strong.
High Melting Point and Boiling Point
Due to very strong force of attraction between ions, ionic compounds have high melting point and high boiling point.
Solubility
Ionic compounds are soluble in water because water and ionic compounds are similar in nature. However, ionic compounds are not soluble in organic solvents like kerosene and petrol.
Electrical Conductivity
The aqueous solution of ionic compounds and molten (fused) ionic compounds conduct electricity because the positive and negative ions are free to move. In the solid state, ionic compounds do not conduct electricity.
Reaction of Sodium and Chlorine
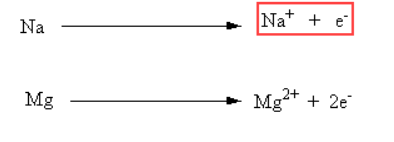
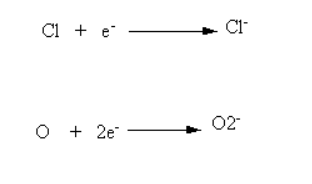
Sodium is a metal. It has a total of 11 electrons.
Its electronic configuration is 2, 8, 1. Because it has one electron in its outermost shell, sodium easily loses this electron. Sodium wants to achieve the electronic configuration of a noble gas by losing one electron.
Chlorine is a non-metal. It has a total of 17 electrons.
Its electronic configuration is 2, 8, 7. Chlorine can gain one electron to complete 8 electrons in its outermost shell.
Reaction of Sodium and Chlorine In the reaction between sodium and chlorine, the sodium atom transfers one electron to the chlorine atom and forms sodium chloride.
Electron transfer process:
Na (2, 8, 1) → Na⁺ (2, 8) + e⁻
Cl (2, 8, 7) + e⁻ → Cl⁻ (2, 8, 8)
After this transfer, Na⁺ and Cl⁻ ions attract each other and form an ionic bond.
Extraction of Metals
The substance from which metals are extracted is called an ore. Those ores from which a metal can be extracted profitably and conveniently are called ores.
The ores which do not allow profitable extraction of metal are called gangue.
All ores are minerals, but all minerals are not ores.
Ores of Metals (Examples and Chemical Formula)
| Metal | Ore | Chemical Formula |
|---|---|---|
| Iron | Hematite | Fe2O3 |
| Iron | Magnetite | Fe3O4 |
| Aluminium | Bauxite | Al2O3 |
| Copper | Copper Sulphide | CuS |
| Zinc | Zinc Sulphide | ZnS |
| Mercury | Cinnabar | HgS |
Various Steps of Extraction of Metals
Concentration of Ore
The ore is crushed and converted into fine powder.
The unwanted impurities present with the ore are separated. This process is called concentration of ore.
The impurities present with the ore are called gangue.
Roasting
Heating the ore in the presence of oxygen is called roasting. This process is used to convert metal sulphides into metal oxides.
Example ZnS + O₂ (heat) → ZnO + SO₂
Calcination
Heating the ore in the absence of oxygen is called calcination. This process is used to convert metal carbonates into metal oxides.
Example ZnCO₃ (heat) → ZnO + CO₂
Corrosion of Metals
The weakening of metals due to environmental factors is called corrosion. The corrosion of iron is called rusting. The rusting of iron is affected by the following factors:
Presence of air
Presence of water
Presence of acidic gases in air
Corrosion of Silver
Silver articles react with sulphur dioxide present in air and form a layer of silver sulphide, which is black in colour.
Corrosion of Copper
Copper reacts with moisture and carbon dioxide to form copper carbonate, which is green in colour.
That is why copper articles turn green after some time.
Reduction
After converting the metal present in the ore into its oxide, it is reduced using carbon. Carbon is used because it is a cheap and easily available reducing agent. Carbon reacts with metal oxide and converts it into pure metal.
Example 2ZnO + C → 2Zn + CO₂ ↑
Extraction of Highly Reactive Metals
Metals like copper, zinc, sodium and potassium are extracted by the electrolytic refining method.
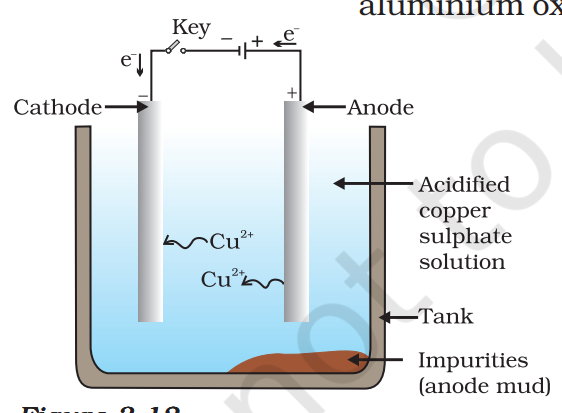
Electrolytic Refining
Anode-Impure metal
Cathode-Pure metal
Electrolyte-Copper sulphate solution with a small amount of sulphuric acid.
Electrolytic Refining (Working Method)
When electric current is passed, the impure metal from the anode dissolves into the solution along with impurities. The pure metal gets deposited on the cathode in the same amount. After some time, all the metal from the anode dissolves and gets deposited on the cathode. The impurities settle down below the anode, which is called anode mud.
Alloy
A homogeneous mixture of two or more metals is called an Alloy
Two important alloys of Copper
Brass– Cu 60-80% + Zinc 40-20%
German Silver – Copper(50)% + Zinc (30)% + Nickel (20)%
Use of Alloys
- Used in making household utensils and wires.
- Used in making statues and idols.











1 comment